G-Protein Coupled Receptors (GPCRs) are one of the largest protein families in the human genome, widely distributed in central nervous system, immune system, cardiovascular system, and retina. As key regulators on the cell membrane, GPCRs transmit external signals into the cell, participating in the regulation of vital physiological activities. Their responses to various endogenous ligands cover a wide range including odors, hormones, and neurotransmitters, showcasing remarkable diversity.
GPCRs are closely related to a variety of diseases, including type II diabetes, obesity, depression, cancer, and Alzheimer's disease. Currently, about 35% of drugs on the market target GPCRs, making them a highly sought-after target for drug development, with significant medical and commercial value. In this context, GPCRs have become an important target for drug development, and their research and drug development have attracted widespread attention.
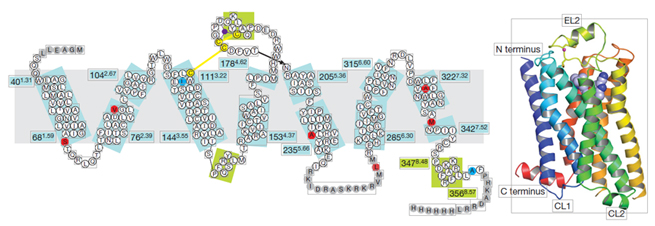
Figure 1. Schematic diagram of GPCR structure
Currently, 481 drugs targeting GPCRs have been approved, accounting for 34% of the total drugs approved by the FDA. About 320 candidate drugs are in clinical trials, with 36% focusing on novel GPCR targets. Among the unapproved drugs, biological drugs, allosteric modulators, and biased agonists are continuously increasing.
The majority of drugs target class A GPCRs (94%), followed by class B (4%), and class C and F each accounting for 2%. Among the marketed drugs, those targeting HRH1, DRD2, ACM1, and ADA1A are the most common, each accounting for more than 5% of the total GPCR-targeted drugs. In terms of disease indications, antihypertensive drugs, antiallergic drugs, analgesics, and antipsychotic drugs are the most widely used. Drugs targeting GPCRs account for approximately 27% of the global market share of therapeutic drugs.
These data indicate that research on GPCRs has never ceased. What makes GPCRs so attractive to many enterprises? Next, we will delve deeper into the related content of GPCRs.
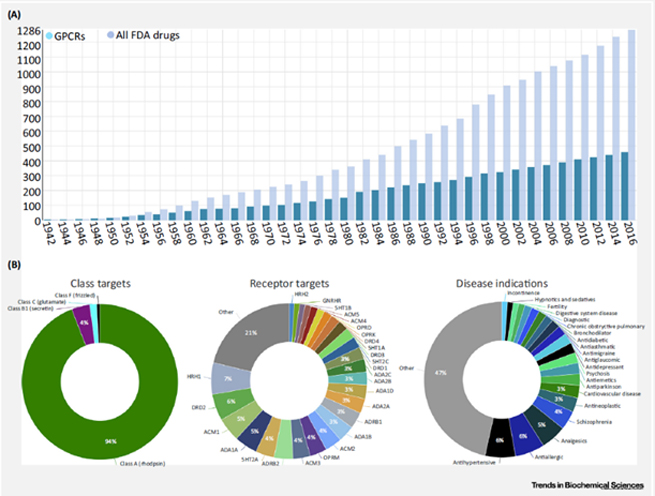
Figure 2. Statistics of G protein-coupled receptor (GPCR) drugs approved by the FDA from 1942 to 2016
GPCRs are receptor proteins with a 7-transmembrane structure, composed of seven α-helical segments, divided into extracellular segments (N-terminal and three Loop structures) and intracellular segments (C-terminal and three Loop structures). The intracellular segments' interaction with downstream G proteins is crucial for signal transduction.
To date, scientists have identified over 800 members of the GPCR superfamily. Due to the family's vastness, there are multiple ways to classify them. Two widely recognized classification methods are: the A-F system, which divides them into six classes based on sequence homology and functional similarity: class A (rhodopsin-like receptors), class B (secretin receptor family), class C (metabotropic glutamate receptors), class D (parasitic mating pheromone receptors), class E (cyclic AMP receptors), and class F (frizzled and smoothened receptors).
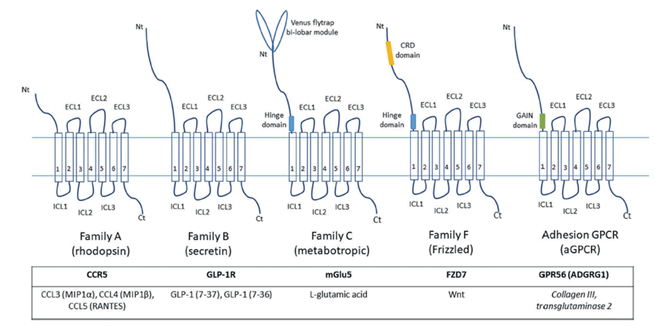
Figure 3. Schematic diagram of key structural features of major GPCR subfamilies
The second is the recently proposed GRAFS classification system based on phylogeny, which includes five main families: Glutamate (G), Rhodopsin (R), Adhesion (A), Frizzled/Taste 2 (F), and Secretin (S).
The trimeric GTP-binding regulatory protein, commonly known as G protein, is composed of α, β, and γ subunits. Each α subunit can independently transmit signals, while β and γ form a heterodimer to function as a whole. The α subunit can be divided into four main families based on function: Gs, Gi/o, Gq/11, and G12/13. These subunits play a key role in signal transduction; for example, Gs and Gi/o can activate or inhibit adenylate cyclase to produce cAMP, which then activates downstream effectors. Gq/11 activates phospholipase Cβ (PLCβ), leading to the hydrolysis of phosphatidylinositol bisphosphate (PIP2) into IP3 and DAG, stimulating the release of Ca2+ from the endoplasmic reticulum. Meanwhile, activated GPCRs are phosphorylated by specific GPCR kinases (GRKs), and β-arrestin is recruited to the phosphorylated GPCR, terminating G protein signaling, leading to receptor desensitization and internalization.
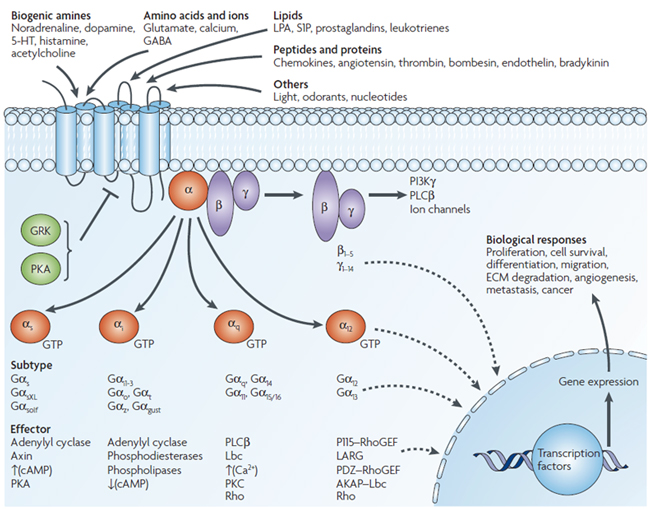
Figure 4. Schematic diagram of the classic signaling pathway of the GPCR Class A family
The cell signaling pathways mediated by G Protein-Coupled Receptors (GPCRs) mainly include the cAMP signaling pathway and the phosphatidylinositol signaling pathway.
In the cAMP signaling pathway, upon binding to external signals, GPCRs can modulate the level of cAMP, converting extracellular signals into intracellular signals. This signaling pathway is further divided into two types based on the type of G protein : the stimulatory G protein receptor, known as the Gs receptor, and the inhibitory G protein receptor, known as the Gi receptor.
In the Gs regulation model, the activated α subunit of Gs activates adenylate cyclase, catalyzing the conversion of ATP to cAMP, which affects the cAMP level and subsequently transmits the signal downstream. The activated βγ subunit complex can also directly activate intracellular target molecules, functioning as a signal transmitter.
Conversely, in the Gi regulation model, the Gi receptor directly inhibits the activity of adenylate cyclase through its α subunit, or by binding the βγ subunit complex to the free α subunit of Gs, blocking its activation of adenylate cyclase, thereby affecting the cAMP level.
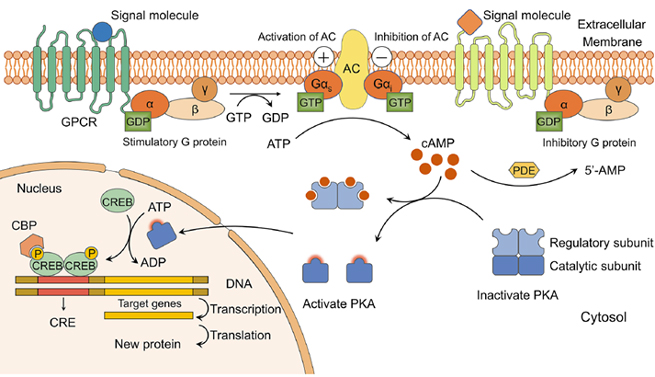
Figure 5. Schematic diagram of the cAMP-PKA signaling pathway
In the phosphatidylinositol signaling pathway, GPCRs bind to external signals, activating PLCβ on the plasma membrane, resulting in the breakdown of PIP2 into IP3 and DAG, the two second messengers. This signaling system is also known as the "double messenger system." IP3 binds to IP3 ligand-gated calcium channels on the endoplasmic reticulum, opening the calcium channels and increasing the cytosolic Ca2+ concentration, activating various Ca2+-dependent proteins. DAG activates PKC, leading to the phosphorylation of serine/threonine residues of proteins, regulating cell functions such as secretion, muscle contraction, cell proliferation, and differentiation. The IP3 signaling pathway is terminated by dephosphorylation or phosphorylation to IP2, IP1, or IP4. Ca2+ is expelled by the plasma membrane pump and retrieved by the endoplasmic reticulum pump. DAG terminates the signal by being enzymatically converted to monoacylglycerol or phosphatidic acid.
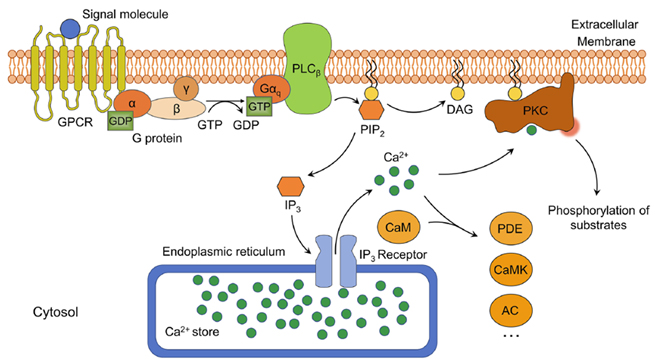
Figure 6. Schematic diagram of the phosphatidylinositol signaling pathway
Currently, about 35% of drugs on the market target GPCRs, making them a significant target for drug development. In the field of drug research and development, establishing GPCR detection methods and ligand screening remains a core focus for scientists. In the past, scientists typically used radioligand binding assays with receptor membranes to screen compounds targeting GPCRs. However, relying solely on binding affinity data is insufficient to determine whether a compound is an agonist or antagonist, and more importantly, this method cannot provide the overall efficacy of the compound under physiological conditions.
Over the past few decades, scientists have been striving to develop signal-dependent cell function analyses to provide more accurate and comprehensive data on compounds targeting GPCRs. This method not only more closely mimics physiological conditions but also allows for a more comprehensive assessment of the compound's impact on GPCRs, thereby providing stronger support for drug development.
An ideal GPCR ligand screening method should be simple, non-radioactive, stable, and homogeneous, and suitable for microplate screening (including 96-well, 384-well, or 1536-well formats) to facilitate automated machine operation. To help you better understand, several widely used GPCR detection methods are listed below.
Receptor binding assays can be used to evaluate the interaction between the receptor and its ligand, such as the intrinsic affinity of the ligand for the receptor, binding/dissociation rates, and the density of the receptor in tissues or cells. Receptor binding assays are a cell-free method theoretically applicable to any GPCRs screening, not involving the receptor's downstream signaling. Traditional radioligand experiments are cumbersome, and even wash-free and filter-free SPA methods still require the use of radioactive ligands, facing challenges with short isotope half-lives and waste disposal. Therefore, emerging non-radioactive methods such as Time-Resolved Fluorescence Resonance Energy Transfer (TR-FRET) technology have been widely promoted, utilizing the long fluorescence lifetime of lanthanide elements like Eu3+ or Tb3+ to detect after all natural fluorescence has disappeared and greatly reducing background fluorescence interference, improving the signal-to-noise ratio.
After ligand binding, GPCRs change their conformation and activate the coupled G protein, which then promotes the generation of second messengers through downstream effectors. Assays to detect G protein activation or G protein-mediated signals, including second messenger generation and reporter gene activation, are referred to as G protein-dependent functional assays. In Figure 1, several highlighted detection indicators are currently the most commonly used, and various mature detection technologies have been developed for different indicators.
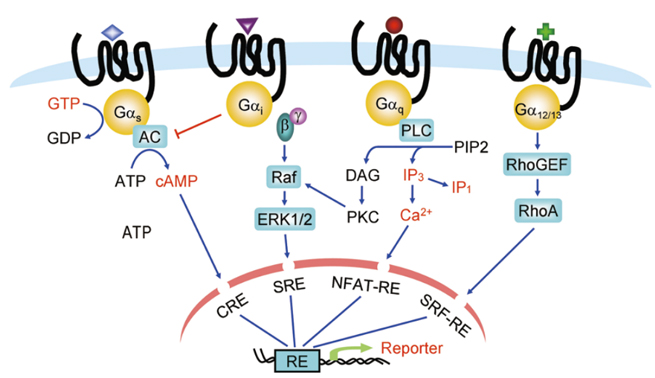
Figure 7. G Protein-Dependent Functional Assays (Highlighted in Red)
The GTPγS binding assay directly measures guanine nucleotide exchange on G proteins, an early event following GPCR activation that is not amplified or modulated by other cellular processes. Following agonist stimulation, the accumulation of non-hydrolyzable GTP analogues (such as [35S]-GTPγS) on the plasma membrane of cells expressing relevant GPCRs can be measured.
Second messengers such as cAMP, IP3/IP1, and Ca2+ are also widely used detection indicators, and many detection methods have been developed. Initially, cAMP and IP3/IP1 were detected using radioactive labeling methods, followed by the development of wash-free SPA methods, and currently, TR-FRET detection schemes have been introduced.
GPCR activation, naturally coupled to Gαq, leads to a increase of Ca2+ ligand-dependent in intracellular.
Using Ca2+ as an indicator to evaluate GPCR activation coupled to Gαi/o, Gαs or Gα12, the signaling pathway can be "switched" by overexpressing chimeric G proteins (Gαqi5 or Gαqo5) or hybrid G proteins (Gα16 or Gα15), inducing an increase of Ca2+ in intracellular . High-throughput screening can be achieved using cell-permeable Ca2+ sensitive fluorescent dyes (such as Fluo-3 and Fluo-4) in conjunction with FLIPR instruments, or by using Ca2+ sensitive biosensors (such as aequorin) to produce a strong luminescent signal.
It is well known that GPCR activation regulates gene transcription through second messenger response elements. These elements include cAMP response elements (CRE), activated T-cell factor response elements (NFAT-RE), serum response elements (SRE), and serum response factor response elements (SRF-RE, a mutant form of SRE). These elements are located in the promoter region of genes and can be detected using reporter gene methods.
Non-G protein-dependent functional assays mainly include GPCR internalization assays, β-arrestin recruitment assays, and label-free functional assays, all of which are based on live cell GPCR functional assays.
GPCR internalization assays are based on receptor desensitization, where GPCRs activated by GRK phosphorylation at specific sites recruit β-arrestin, leading to receptor endocytosis. With the rise of image-based high-content screening (HCS) systems, GPCR internalization detection has become quantifiable.
β-arrestin recruitment is a common mechanism of negative regulation of GPCR signaling, applicable to almost all GPCRs. For screening drugs related to Gαi-coupled GPCRs and orphan GPCRs, the detection window of traditional second messenger detection systems is small, making β-arrestin recruitment detection methods particularly useful. Various non-imaging-based β-arrestin recruitment detection methods have been developed, including bioluminescence resonance energy transfer (BRET), TangoTM detection (Invitrogen), and PathHunterTM technology (DiscoveRx).
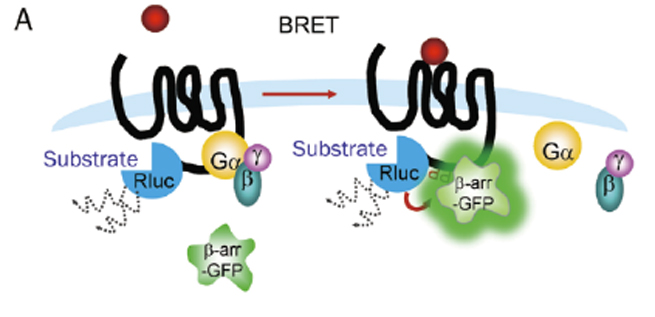
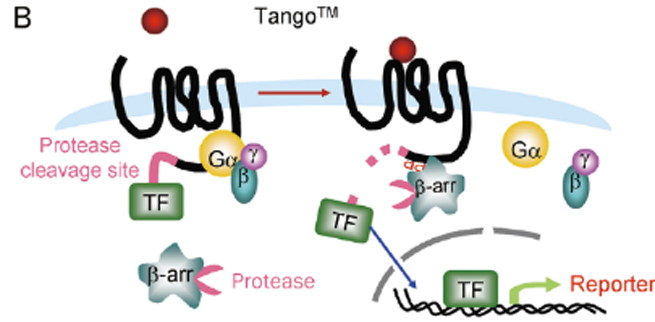
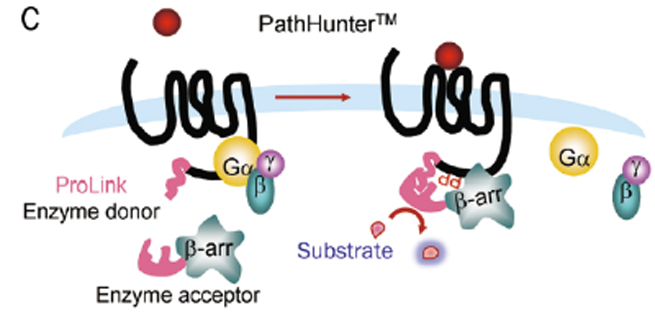
Figure 8. β-arrestin Recruitment Detection Methods
(A. BRET; B. TangoTM; C. PathHunterTM)
Label-free technology has emerged in the field of whole-cell assays, especially in GPCR screening. This method uses biosensors to convert ligand-induced changes into quantifiable signals such as optics or electricity, thereby integrating cellular responses. Label-free whole-cell assays can monitor changes in cell characteristics, including adhesion, proliferation, migration, and cell death. The main instruments currently used include BINDTM and EpicTM (optical) as well as ECISTM, xCELLigenceTM, and CellKeyTM (electrical). Despite challenges with false positives and negatives, label-free technology, when combined with traditional methods, is expected to further expand the application of GPCRs in drug discovery and development.
Many GPCRs form dimers on the plasma membrane, which has profound implications for signal transduction and pharmacology. C-type GPCRs require heterodimerization to maintain function. Many A-type GPCRs can form homodimers when expressed alone, and their heterodimerization may lead to different signaling characteristics. Methods have been designed to assess the effects of compounds on GPCR dimerization, and technologies established include FRET/BRET, Tag-liteTM, and PathHunterTM GPCR dimerization detection systems.

Figure 9. Receptor Dimerization Assays
(A. BRET; B. Tag-liteTM; C. PathHunterTM)
GPCRs activate G proteins to transmit external stimuli, triggering signal cascades. In this process, G proteins replace GDP with GTP, causing the α and β/γ subunits to dissociate and trigger different signaling pathways. Different kinases, such as Src, PLCβ, PKC, etc., regulate small G protein/MAPK cascades. Detecting phosphorylated protein levels is crucial for understanding this process. From the initial radioactive labeling to phosphorylation-specific antibodies, various immunoassay techniques have been promoted. Such as Western Blot, ELISA, flow cytometry. Among them, TR-FRET detection technique is a simple and homogeneous method, which is suitable for high throughput screening.